Translate this page into:
Prevalence of pre-operative anemia and its implication for patient blood management in a Nigerian Hospital

-
Received: ,
Accepted: ,
How to cite this article: Kalu Q, Usoro N, Etta O, Eshiemomoh R, Enabulele J. Prevalence of pre-operative anemia and its implication for patient blood management in a Nigerian Hospital. Calabar J Health Sci 2022;6:59-64.
Abstract
Objectives:
Pre-operative anemia is common in Sub-Saharan Africa including Nigeria. Anemia is a known risk factor for blood transfusion. In our hospital, like many others, patients are usually admitted a day before elective surgeries and the default treatment for anemia by most surgeons and anesthetists is allogenic blood transfusion. Anemia and blood transfusion are independently and synergistically associated with complications and undesirable outcomes. Patient blood management (PBM) is gaining popularity and has been advocated by the World Health Organization (WHO) for member countries to adopt as a means of improving patient outcomes. There is currently no research from the University of Calabar Teaching Hospital highlighting the prevalence of pre-operative anemia. This research was aimed at filling this gap and also presenting a baseline for comparison as the hospital strives to implement the three pillars of PBM strategies: Manage anemia, minimize blood loss and harness tolerance to anemia to improve patient outcomes.
Material and Methods:
All surgical patients who consented to the study were recruited prospectively. Patients who had no pre-operative hemoglobin (Hgb) or packed cell volume (PCV) recorded were excluded from the study. Demographic data, type of surgery, and pre-operative PCV levels were documented. Anemia was defined using the WHO standard as PCV <39% (Hgb 13.0 g/dL) in men and <36% (Hgb 12.0 g/dL) in women. Data were obtained about transfusion at the end of surgery. Surgeries were categorized into general, orthopedic, obstetric, gynecological, urologic, pediatric, ear, nose, throat/maxillofacial, burns and plastic unit (BPU), neurological, and thoracic. The information was entered into SPSS version 20. Data were cleaned and analyzed. The statistical significance was placed at P < 0.05. Results are presented as tables, chats, and histograms.
Results:
A total of 237 patients were recruited with a mean age of 37.30 ± 19.05. More of them were females (54.0%) and a higher proportion of them (64.6%) had elective surgery. The categories of surgeries were predominantly general surgical (24.9%) and gynecological (21.9%) followed by orthopedic (17.7%), ENT/maxillofacial (9.3%), urologic (8.0%), and obstetric (7.2%) cases. The rest were neurosurgical (3.4%) and pediatric surgical (2.1%) cases. The overall prevalence of anemia was found to be 54.9%. The mean pre-operative PCV was 34.90 ± 7.37%. The highest proportion of pre-operative anemia was found among the pediatric surgical cases (80.0%) followed by the urologic (68.4%) and neurosurgical cases (62.5%). The prevalence of pre-operative anemia among the different surgical patients was gynecological (46.2%), general surgical (45.8%), obstetric (41.2%), ENT/maxillofacial (40.9%), and orthopedic (38.1%) cases. The lowest proportion of pre-operative anemic patients was found among the thoracic (25.0%) and Burns and Plastics (11.1%) cases. Most of the patients (86.5%) were not transfused intraoperatively. Whereas 20% of the anemic patients were transfused; only 5.6% of the non-anemic ones needed a blood transfusion.
Conclusion:
Pre-operative anemia remains prevalent in our environment and anemia is a risk factor for a blood transfusion which carries both risk and cost implications. PBM has as its first pillar the management of anemia. This pre-emptive approach to allogeneic blood transfusion avoidance is aimed at improving patient outcomes. Prevention and treatment of pre-operative anemia will be beneficial not just to the patients but to all healthcare stakeholders.
Keywords
Pre-operative
Anemia
Patient blood management
Nigeria
INTRODUCTION
According to the World Health Organization (WHO), anemia is defined as hemoglobin (Hgb) concentration <13.0 g/dL in men and <12.0 g/dL in women.[1] Anemia resulting in tissue ischemia remains a dreaded condition. Many patients receive blood transfusions every day in hope of avoiding the negative consequences of anemia including tissue ischemia.[2-4] Researchers are recently, however, drawing clinicians’ attention to the need for a balance between the risks of transfusion and risks of anemia as well as the challenges of inadequate education and information regarding this subject.[1] A standardized approach for the detection, evaluation and management of pre-operative anemia in our resource-challenged setting is an identified unmet medical need. The burden of pre-operative anemia is also largely undocumented. There is currently no institutional Hgb “transfusion trigger” in our setting, thereby leaving allogeneic blood transfusion (ABT) decisions to the discretion of the individual surgeon and anesthetist.
Patient blood management (PBM) is defined by the WHO as a patient-centered approach that addresses iron deficiency, anemia, coagulopathy, and blood loss in both surgical and non-surgical patients, as risk factors for adverse medical outcomes.[5] The aim of PBM as highlighted by the WHO is to improve patient outcomes while saving healthcare resources and this was endorsed by the World Health Assembly resolution WHA 63.12(140).[5] Blood transfusion addresses the administration of allogeneic or autologous blood and blood components while adjuvant therapies refer to drugs and techniques to reduce or prevent blood loss and the need for transfusion of allogeneic blood.[6] Pre-operative anemia with an increased likelihood of blood transfusion is common in elective surgical patients in the West African subregion.[3] Transfusion is associated with several complications including the transmission of bloodborne diseases, immunomodulation, transfusion-associated circulatory overload, transfusion-associated lung injury (TRALI), and others. Anemia itself is an independent risk factor for perioperative morbidity and mortality.[2] This includes myocardial and other tissue ischemia, and wound infections which may necessitate unplanned intensive care unit admissions or prolonged hospital admission. Patients are very often booked for surgery and admitted a day before the scheduled operation in our hospital facilities in Nigeria and other similar countries in Sub-Saharan Africa. This, therefore, leaves little or no time for pre-operative optimization of the anemic patient. Anesthetists and surgeons prepare for and resort to ABT once the allowable blood loss is exceeded. The restrictive as well as goal-directed fluid including blood management is known by anesthetists. In practice, however, it appears that the liberal fluid management approach is most popular.
This study was aimed at establishing the prevalence of pre-operative anemia among patients presenting for non-cardiac surgeries at the University of Calabar Teaching Hospital (UCTH), Nigeria because the prevalence of pre-operative anemia in the hospital is currently unknown and not published. This knowledge will serve as a baseline for the assessment of the effectiveness of specific PBM strategies when they become fully implemented.
MATERIAL AND METHODS
The study was carried out in the UCTH in South Nigeria. The hospital is a tertiary health-care facility that serves as a training institution for medical students, postgraduate medical doctors, and allied medical personnel. It has many surgical specialties engaged in minor and major surgeries including obstetrics and gynecology, orthopedics, pediatric, urologic, neurological, dental, maxillofacial, general, burns and plastic surgeries, and among many others. Following approval by the Health Research and Ethics Committee of the Hospital and informed consent by participants, consecutive patients who consented to the study were recruited prospectively. Patients who had no pre-operative Hgb or packed cell volume (PCV) recorded were excluded from the study. A data sheet was used to obtain demographic data, type of surgery, and pre-operative PCV levels and whether the patient was transfused or not was recorded postoperatively. Anemia was defined as PCV <39% (Hgb 13.0 g/dL) in men and <36% (Hgb 12.0 g/dL) in women.[1] In this study, PCV was used based on the assertion that Hgb is estimated as a third of the PCV.[7]
Surgeries were categorized into general, orthopedic, obstetric, gynecological, urologic, pediatric, ear, nose, throat/maxillofacial, burns and plastic (BPU), neurological, and thoracic. The information was entered into SPSS version 20. Data were cleaned and analyzed and the statistical significance was placed at P < 0.05. A Chi-square test of independence was conducted to explore the relationship between pre-operative anemia, type of surgery, categories of surgeries, and the need for transfusion. Results are presented as tables, chats, and histograms.
RESULTS
Data were obtained from a total of 237 consecutive patients who consented to the study and met the inclusion criteria. The mean age of the patients was 37.30 ± 19.05. The majority were within the age group of 21–40 years (42.6%), followed by 41–60 years (28.7%) then 1–20 years (16.9%) and 61– 80 years (11.0%). Only 0.8% of patients were above 80 years.
More of them were female (54.0%) and a higher proportion of them (64.6%) had elective surgery. The majority of the patients underwent general surgery (24.9%) and gynecological procedures (21.9%), while the remaining had orthopedic (17.7%), ENT/maxillofacial (9.3%), urologic (8.0%), obstetric (7.2%), neurologic (3.4%), and pediatric surgeries (2.1%). The mean pre-operative PCV was 34.90 ± 7.37 %. Most of the patients (86.5%) were not transfused intraoperatively.
Relationship between pre-operative anemia, type of surgery and categories of surgeries, and the need for intraoperative transfusion
The overall prevalence of anemia was found to be 54.9%. Figure 1 shows that 55% of males and 54.7% of females were anemic preoperatively. As shown in [Figure 2], the highest proportion of pre-operative anemia was found among the pediatric surgical cases (80.0%) followed by the urologic (68.4%) and neurosurgical cases (62.5%).
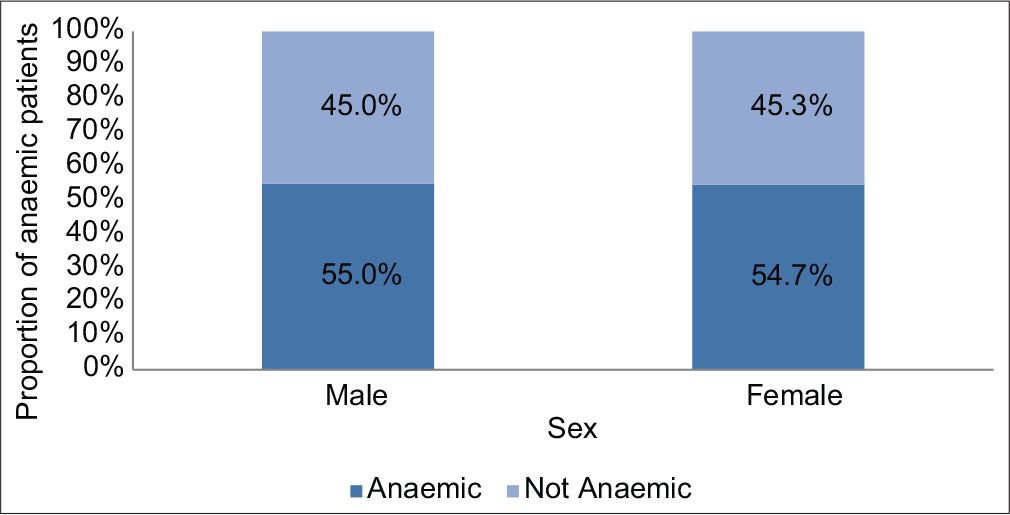
- A composite bar chart showing the proportion of patients admitted for surgery in UCTH, Calabar, that were anemic by sex before surgery.
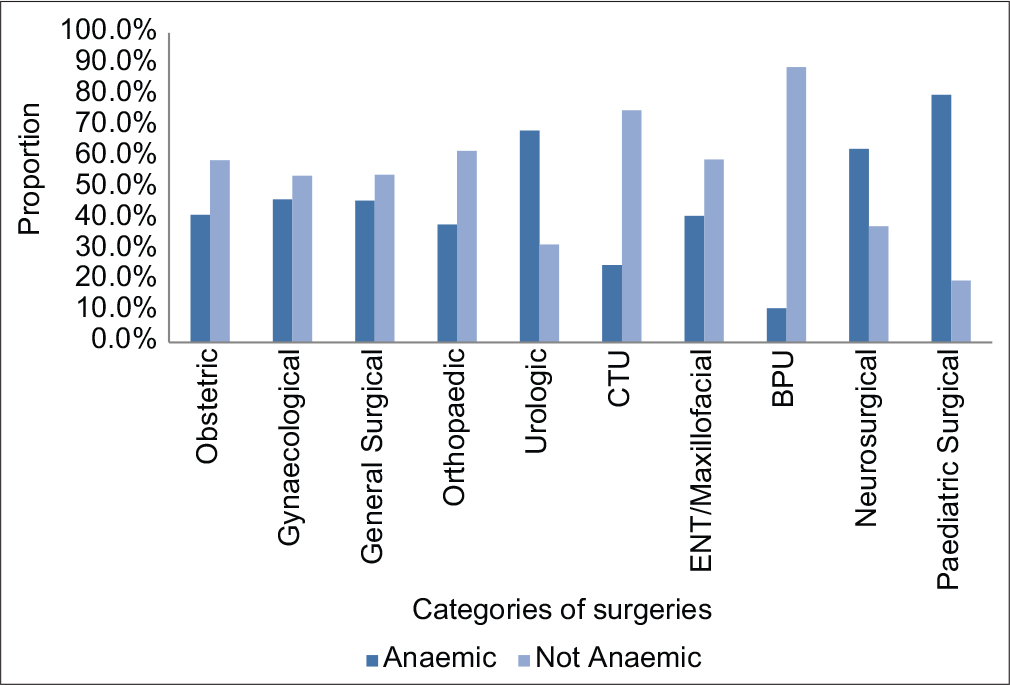
- A multiple bar chart showing the proportions of patients who were anemic across the different categories of case diagnosis before surgery in UCTH, Calabar.
The lowest proportion of pre-operative anemic patients was found among the thoracic (CTU) (25.0%) and BPU (11.1%) cases while gynecological patients had 46.2%, general surgical (45.8%), obstetric (41.2%), ENT/maxillofacial (40.9%), and orthopedic (38.1%) cases. The sociodemographic and clinical characteristics of patients are shown in Table 1.
| Variable | Mean±SD | Frequency | Percentage |
|---|---|---|---|
| Age | 37.30±19.05 | ||
| Age group | |||
| 1–20 | 40 | 16.9 | |
| 21–40 | 101 | 42.6 | |
| 41–60 | 68 | 28.7 | |
| 61–80 | 26 | 11.0 | |
| >80 | 2 | 0.8 | |
| Sex | |||
| Male | 109 | 46.0 | |
| Female | 128 | 54.0 | |
| Categories of surgeries | |||
| Obstetrics | 17 | 7.2 | |
| Gynecology | 52 | 21.9 | |
| General surgery | 59 | 24.9 | |
| Orthopedics | 42 | 17.7 | |
| Urology | 19 | 8.0 | |
| CTU | 4 | 1.7 | |
| ENT/maxillofacial | 22 | 9.3 | |
| BPU | 9 | 3.8 | |
| Neurosurgery | 8 | 3.4 | |
| Pediatric surgery | 5 | 2.1 | |
| Type of surgery | |||
| Emergency | 84 | 35.4 | |
| Elective | 153 | 64.6 | |
| Unit of blood transfused | |||
| Nil | 205 | 86.5 | |
| 1 | 8 | 3.4 | |
| 2 | 20 | 8.4 | |
| 3 | 4 | 1.7 | |
| Pre-operative PCV (%) | 34.90±7.37 |
SD: Standard deviation
There was a significant relationship between the categories of surgeries and the need for blood transfusion (P < 0.005). The areas in greatest need of blood transfusion were the gynecological cases (30.8%), followed by general surgical (15.3%), obstetric (11.8%), orthopedic (9.5%), and urologic (5.3%) cases but there was no need for transfusion in the other categories of case diagnosis, namely, CTU, ENT/ maxillofacial, BPU, neurosurgical, and pediatric surgical cases. However, there was no significant association between the type of surgery patients booked for (i.e., emergency or elective) and the need for intraoperative transfusion (P = 0.794), as shown in [Table 2].
| Factor | Transfused? | Chisquare test |
P-value | |
|---|---|---|---|---|
| Yes (%) | No (%) | |||
| Pre-operative anemia | ||||
| Yes | 26 (20.0) | 104 (80.0) | 10.41 | 0.001* |
| No | 6 (5.6) | 101 (94.4) | ||
| Type of surgery | ||||
| Emergency | 12 (14.3) | 72 (85.7) | 0.07 | 0.794 |
| Elective | 20 (13.1) | 133 (86.9) | ||
| Categories of surgeries | ||||
| Obstetrics | 2 (11.8) | 15 (88.2) | 14.02 | <0.005* |
| Gynecology | 16 (30.8) | 36 (69.2) | ||
| General surgery | 9 (15.3) | 50 (84.7) | ||
| Orthopedics | 4 (9.5) | 38 (90.5) | ||
| Urology | 1 (5.3) | 18 (94.3) | ||
| CTU | 0 (0.0) | 4 (100.0) | ||
| ENT/maxillofacial | 0 (0.0) | 22 (100.0) | ||
| BPU | 0 (0.0) | 9 (100.0) | ||
| Neurosurgery | 0 (0.0) | 8 (100.0) | ||
| Pediatric surgery | 0 (0.0) | 5 (100.0) | ||
*Significant P value
DISCUSSION
The aim of this paper was to find the prevalence of pre-operative anemia generally and in different groups of surgical patients presenting for non-cardiac surgical procedures. The hospital has a PBM committee which is newly formed. It is hoped that with this committee in place, a uniform blood transfusion trigger will be adopted which is not the case currently. Pre-operative anemia in this study was 54.9%. This is similar to the finding by Amponsah in Ghana who reported a prevalence of 54.3% among non-cardiac patients.[3] This similarity may be because the patient population is similar and Ghana, like Nigeria, is a malaria-endemic region. Some authors have suggested that anemia could be an adaptation to living in malaria-endemic regions.[8] In contrast, Brisbe et al. found a prevalence of 10.5% in their series.[9] However, unlike ours, only patients billed for orthopedic surgery were recruited in their study and, additionally, their geographical location is in the Western world where malaria is not endemic. There was no significant difference in male/female prevalence even though the majority of the patients were in the reproductive age bracket. This finding is similar to the WHO study findings.[1] The fact that the definition of anemia already takes the male/female difference into cognizance may explain this. Shander et al. in their study also found no difference in anemia prevalence between male and female participants.[10] Their study was a systematic review of the literature involving many other studies. A study in Singapore, however, found that females aged 18–70 years had a significantly higher prevalence of anemia than males.[11] This was attributed to menstruation and pregnancy. The same study, however, found the trend to reverse in older patients.
This study was a retrospective study and, as such, it is possible that many confounding variables were not considered. The National Health and Nutrition Examination Survey 111 in the United States of America found a significantly higher anemia prevalence among females compared to males[12] but their study was among the general population unlike the pre-operative patients evaluated in this work.
About 80% of the pediatric surgery patients in this study were anemic. This finding is similar to studies by the WHO with an anemia prevalence of over 70% among pediatric patients in low and middle income countries (LMIC).[13] This high prevalence has been attributed to malnutrition, parasitic infections, hemoglobinopathies, and HIV and AIDS. Malnutrition remains a big problem in Sub-Saharan Africa with consequent iron deficiency worsened by malaria infestation. Yang et al. had a lower prevalence of 40% in their study involving 24 LMICs. Several socioeconomic discrepancies were highlighted as responsible.[14] The prevalence in developed countries, on the other hand, is a sharp contrast. Faraoni et al.[15] found a 24% prevalence which was significantly associated with an increased risk of in-hospital mortality after adjusting for comorbidities. This highlights the need for management strategies for pre-operative anemia because timely detection and intervention, especially in the context of elective surgery, can theoretically reduce the need for transfusions. In LMICs, there are additional concerns about blood transfusion safety and access-related issues.[16]
Urology patients had a 68.4% prevalence of preoperative anemia. This is not surprising although further investigations are needed to confirm anemia of chronic renal disease. Other studies indicate that patients scheduled for major urological cancer surgery are likely to have iron deficiency, with or without anemia with a reported prevalence of pre-operative anemia for patients undergoing radical nephrectomy at 35% and nephroureterectomy at 39.7%.[6,17,18] Interestingly, Munoz et al. found a prevalence of 8% among patients presenting for radical prostatectomy.[19] The reason for this low prevalence is not clear but the use of erythropoietin and parenteral iron has been shown to correct anemia of chronic renal disease in several studies. Chronic renal disease interferes with hematopoiesis. The severity of anemia of chronic renal disease is directly related to the degree of loss of kidney function, as the kidneys are responsible for approximately 90% of erythropoietin production.[17] The etiology of anemia in these patients is multifactorial and includes decreased red blood cell production due to lack of erythropoietin and iron deficiency, increased red blood cell destruction due to hemolysis, and increased blood loss due to multiple phlebotomies for an array of indications.[18]
Pre-operative anemia was significantly related to ABT in this study. This finding is similar to that of several other authors.[20-22] The advocacy for blood transfusion avoidance has significantly reduced ABT in our hospital. In this series, with 54.9% of anemic patients presenting for surgery, only 13.5% were transfused. The majority of the transfusions were for gynecological surgery. Blood transfusion is largely a treatment option in severe anemia. The degree of anemia was not ascertained in most cases and the estimated blood loss was not recorded. Point-of-care Hgb measurement is not a pre-requisite for blood transfusion in our center. Clinical judgment often comes into play in the decision to transfuse or not. The cost barrier is a possible reason why some anemic patients may not be transfused. This is because our healthcare access is mostly through the “fee-for-service” arrangement. In contrast, ABT is one of the top five overused procedures in the United States of America.[23] Findings from other authors have shown that obstetrics, urology, thoracic, and major orthopedic surgery such as total knee replacement and total hip replacement are associated with high ABT rates.[11,24-26] PBM is gradually gaining popularity in our center with its three pillars being: (1) Detection and management of anemia and iron deficiency, (2) minimization of blood loss and optimization of coagulation, and (3) leveraging and optimizing the patient’s specific physiological tolerance of anemia.[5] Effective intervention in our setting will be to detect and manage anemia and iron deficiency. As this study shows, less anemia means less allogenic blood transfusion. Anemia should be a reason to delay elective surgery and buy time for the patient’s optimization rather than the immediate option of blood transfusion. Effective optimization of pre-operative hematocrit is achievable through the pre-anesthesia and surgery screening clinic. Anemic patients should be referred to this clinic for corrective interventions. Hgb and PCV should be measured as soon as surgery is scheduled. This is recommended to be 4–8 weeks before surgery. Allowable blood loss should be calculated routinely.
Minimizing blood loss involves teamwork and the surgical safety checklist should be administered. This allows team members to know the anticipated blood loss, whether antibiotics have been given and the estimated duration of surgery, among others. It enables every team member to know when things go wrong. Reduced perioperative phlebotomy, proper positioning to minimize blood loss, use of hypotensive anesthesia where indicated, maintenance of normothermia, intraoperative blood salvage, normovolemic hemodilution, use of tourniquets, diathermy, surgical sealants, and monitoring coagulation are some methods of minimizing blood loss. Optimizing oxygenation is achieved by increasing the cardiac output, oxygen therapy, mechanical ventilation, and other methods of increasing oxygen delivery while minimizing consumption. The last pillar is the lowering of the transfusion trigger which emphasizes the need to manage the patient and not just the Hgb value. Intraoperative Hgb monitoring is possible using point-of-care testing like the hemocue. Management of patients for whom ABT is not an option has revealed that tolerance of acute anemia to incredibly low levels is possible.
The limitations of this study were that patients were recruited consecutively irrespective of the type of non-cardiac surgery. Accordingly, some surgical specialties had more patients than others. Furthermore, the causes of anemia such as iron deficiency, inflammation, and chronic diseases, among others, were not determined. In addition, the study design did not capture the type of anesthesia, estimated blood loss, and consequences of anemia such as prolonged hospital stay and unplanned intensive care unit admission. Other areas for future research in our facility will include the other two PBM pillars; minimization of blood loss and optimization of coagulation and leveraging and optimizing the patient’s specific physiological tolerance of anemia. Accordingly, the effectiveness of our scoop and sieve method of intraoperative blood salvage and normovolemic hemodilution technique of autologous blood transfusion will be studied scientifically for future publication.
CONCLUSION
Pre-operative anemia remains prevalent in our environment and anemia is a risk factor for a blood transfusion which carries both risk and cost implications. PBM has as its first pillar the management of anemia. This pre-emptive approach to ABT avoidance is aimed at improving patient outcomes. Prevention and treatment of pre-operative anemia will be beneficial not only to the patients but also to all healthcare stakeholders.
Declaration of patient consent
Informed consent was obtained from each participant even though their identity were neither disclosed nor compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Haemoglobin Concentrations for the Diagnosis of Anaemia and Assessment of Severity. 2017. Geneva: World Health Organization; Available from: http://www.WHO_NMH_NHD_MNM_11.1_eng.pdf [Last accessed on 2022 Apr 20]
- [Google Scholar]
- What is really dangerous: Anaemia or transfusion. Br J Anaesth. 2011;107:141-59.
- [CrossRef] [PubMed] [Google Scholar]
- Preoperative anaemia and associated postoperative outcomes in non-cardiac surgery patients in Central Region of Ghana. Anaesthesiol Res Pract. 2017;2017:7410960.
- [CrossRef] [PubMed] [Google Scholar]
- Anemia and its clinical consequences in patients with chronic diseases. Am J Med. 2004;116(Suppl 7A):1S-2.
- [CrossRef] [PubMed] [Google Scholar]
- The Urgent Need to Implement Patient Blood Management: Policy Brief. 2022. Geneva: World Health Organization; Available from: https://www.apps.who.int/iris/handle/10665/346655 [Last accessed on 2022 Apr 20]
- [Google Scholar]
- International consensus statement on the perioperative management of anaemia and iron deficiency. Anaesthesia. 2016;72:233-47.
- [CrossRef] [PubMed] [Google Scholar]
- Basic haematological techniques In: Lewis SM, Bain BJ, Bates I, eds. Practical Haematology (9th ed). Edinburgh: Churchill Livingstone; 2001. p. :19-46.
- [Google Scholar]
- Anemia, iron supplementation and susceptibility to Plasmodium falciparum malaria. EBioMedicine. 2016;14:13-4.
- [CrossRef] [PubMed] [Google Scholar]
- Prevalence of preoperative anemia and hematinic deficiencies in patients scheduled for elective major orthopedic surgery. Transfus Alter Transfus Med. 2008;10:166-73.
- [CrossRef] [Google Scholar]
- Prevalence and outcomes of anemia in surgery: A systematic review of the literature. Am J Med. 2004;16(Suppl 7A):58S-69.
- [CrossRef] [PubMed] [Google Scholar]
- Prevalence of preoperative anemia, abnormal mean corpuscular volume and red cell distribution width among surgical patients in Singapore, and their influence on one year mortality. PLoS One. 2017;12:e0182543.
- [CrossRef] [PubMed] [Google Scholar]
- The prevalence of anemia and moderate-severe anemia in the US population (NHANES 2003-2012) PLoS One. 2016;11:e0166635.
- [CrossRef] [PubMed] [Google Scholar]
- Surgical decision making around paediatric preoperative anaemia in low-income and middle-income countries. Lancet Child Adolesc Health. 2019;3:814-21.
- [CrossRef] [PubMed] [Google Scholar]
- Trends in socioeconomic inequalities and prevalence of anemia among children and nonpregnant women in low and middle-income countries. JAMA Netw Open. 2018;32:e182899.
- [CrossRef] [PubMed] [Google Scholar]
- Relationship between preoperative anemia and in-hospital mortality in children undergoing noncardiac surgery. Anesth Analg. 2016;123:1582-7.
- [CrossRef] [PubMed] [Google Scholar]
- Preoperative anemia increases the risk of red blood cell transfusion and prolonged hospital length of stay in children undergoing spine arthrodesis surgery. Transfusion. 2019;59:492-9.
- [CrossRef] [PubMed] [Google Scholar]
- The prevalence of anemia in patients with chronic kidney disease. Curr Med Res Opin. 2004;20:1501-10.
- [CrossRef] [PubMed] [Google Scholar]
- Iron deficiency in chronic kidney disease: Updates on pathophysiology, diagnosis, and treatment. J Am Soc Nephrol. 2020;31:456-68.
- [CrossRef] [PubMed] [Google Scholar]
- Pre-operative haemoglobin levels and iron status in a large multicentre cohort of patients undergoing major elective surgery. Anaesthesia. 2017;72:826-34.
- [CrossRef] [PubMed] [Google Scholar]
- British committee for standards in haematology guidelines on the identification and management of pre-operative anaemia. Br J Haematol. 2015;171:322-31.
- [CrossRef] [PubMed] [Google Scholar]
- Management of perioperative iron deficiency anemia. Acta Haematol. 2019;142:21-9.
- [CrossRef] [PubMed] [Google Scholar]
- Impact of preoperative erythropoietin on allogeneic blood transfusions in surgical patients: Results from a systematic review and meta-analysis. Anesth Analg. 2019;128:981-92.
- [CrossRef] [PubMed] [Google Scholar]
- Transfusion Overuse Exposing an International Problem and Patient Safety Issue. Available from: https://www.sabm.org/wp-content/uploads/2018/08/transfusion-overuse.pdf [Last accessed on 2020 Sep 29]
- [Google Scholar]
- Detection, evaluation, and management of preoperative anaemia in the elective orthopaedic surgical patient: NATA guidelines. Br J Anaesth. 2011;106:13-22.
- [CrossRef] [PubMed] [Google Scholar]
- Pre-operative anaemia: Prevalence, consequences and approaches to management. Blood Transfus. 2015;13:370-9.
- [Google Scholar]
- Preoperative screening and intervention for mild anemia with low iron stores in elective hip and knee arthroplasty. Transfusion. 2017;57:3049-57.
- [CrossRef] [PubMed] [Google Scholar]






