Translate this page into:
Evaluation of microbial flora of the external surface of housefly (Musca domestica) in Umuahia Metropolis, Abia State, Southeast Nigeria

*Corresponding author: E. O. Nwankwo, Department of Microbiology, College of Natural Sciences, Michael Okpara University of Agriculture, Umudike, Umuahia, Abia State, Southeast Nigeria. emmaonwubiko@yahoo.com
-
Received: ,
Accepted: ,
How to cite this article: Nwankwo EO, Ekemezie CL, Adeyemo S. Evaluation of microbial flora of the external surface of housefly (Musca domestica) in Umuahia Metropolis, Abia State, Southeast Nigeria. Calabar J Health Sci 2019;3(1):9-15.
Abstract
Objective:
Houseflies are vectors responsible for the mechanical transmission of pathogens acquired from feeding in feces and decayed organic debris. Human consumption of such food without warming could lead to gastroenteritis, a major public health problem. The aim of this research was to evaluate the range of microbial pathogens associated with the external surfaces of fly vectors and to determine the antibiotic susceptibility pattern of the bacterial pathogens.
Materials and Methods:
A total of 150 houseflies were collected with a sterile net from different parts of Umuahia, Abia State. Their external surfaces were screened for bacteria, fungi, and protozoan parasites in the Microbiology Laboratory of Michael Okpara University of Agriculture, Umudike by standard microbiological procedures. Antibiotic sensitivity pattern of bacterial isolates was carried out by disc diffusion method.
Results:
The most frequently observed microorganisms were Escherichia coli (22.9%), Klebsiella spp. (16.6%), Staphylococcus aureus (14.6%), Aspergillus spp. (28.3%), Mucor spp. (21.7%), Entamoeba histolytica (32.7%), and Endolimax nana (30.9%). Houseflies from broken sewage had the highest total viable counts and frequency of bacteria, fungi, and parasites. Bacterial isolates from houseflies gotten from health-care facilities showed higher levels of multiple drug resistance to ampicillin and cotrimoxazole.
Conclusion:
In this study, pathogenic microorganisms were recovered from the external surface of houseflies, the vectoral agents of mechanical transfer of microbial contaminants to exposed food. Most of the microorganisms observed in this study are known pathogens that can cause gastroenteritis which is a public health concern.
Keywords
Antibiotic susceptibility
External body surface
Houseflies
Microbial vectors
INTRODUCTION
Houseflies (Musca domestica) are common insects of the family Muscidae order Diptera. They are synanthropic insects that are widely distributed globally. They enter into several places, including contaminated premises due to their own biologic habits of feeding. The habits of housefly favor the spread of bacteria and other diseases causing organisms. Consequently, housefly, for example, can spread diseases such as food poisoning and dysentery.[1] The behavioral characteristics of the housefly, M. domestica, ensure its contact with food and wastes of man and animals and in this manner are able to transport pathogenic organisms from contaminated materials to man.[2]
It is a vector responsible for the mechanical transmission of pathogens borne on its body parts acquired from feeding on feces and decayed organic debris.[3,4] The isolation of pathogenic bacteria from the feces of houseflies has proved the transmission by fecal oral route as feasible.[2] In addition to their role in disease transmission, houseflies are usually regarded as indicator agents, symbolic of disposal problems and reflecting the sanitary level of the community in the absence of valid statistical data, and bacteriological information about an essential health situation.[5]
M. domestica is a medically important insect implicated in the transmission of various human pathogens such as Vibrio cholerae, Enterobacteriaceae, Staphylococcus aureus, Pseudomonas spp., Shigella spp., Salmonella spp., rotavirus, eggs of metazoa, and protozoan cysts.[6] They are the major epidemiologic factors responsible for the spread of acute gastroenteritis, trachoma among infants and young children in developing countries, and transmission of nosocomial infections with multiple antibiotic-resistant bacteria.[7]
Structurally, the fly is well adapted for picking up pathogens. Its proboscis is provided with a profusion of fine hairs that readily collect environmental debris. Furthermore, each of the six legs of the fly is fitted with hairy structures and pads that secrete a sticky material, thus adding to its pathogen transmission potential.[2]
The aim of this research was to evaluate the range of microbial pathogens associated with the external surfaces of houseflies and to determine the antibiotic susceptibility pattern of recovered bacterial pathogens.
MATERIALS AND METHODS
Sample collection
A total of 150 houseflies were collected from five different sites with sterile sweep net, namely, refuse dump sites, broken sewage, restaurants, relief market, and school clinic all located within Michael Okpara University of Agriculture, Umudike. An average of 30 houseflies was collected from each site between the hours of 8.00 am and 10.00 am and placed in sterile containers for analysis. Their external surfaces were processed by standard microbiological procedures[8] and screened for bacteria, fungi, and intestinal parasites.
Determination of total viable counts
Five houseflies each were placed into a sterile clean universal container containing 2 ml of physiological saline solution, shaken vigorously for about 2 min and the flies aseptically removed from the solution. The resulting suspension was serially diluted by 10- fold and 0.1 ml of each dilution cultured on nutrient agar plates and incubated aerobically at 37°C for 24 h.
Culture
Organisms that grew on the nutrient agar were later subcultured into blood agar, MacConkey agar, and Mannitol salt agar, respectively, for bacterial isolation. Sabouraud dextrose agar was used for the isolation of fungal pathogens. While culture plates for bacterial isolation were incubated aerobically at 37°C for 18–24 h, fungal culture plates were incubated at room temperature for 2–3 days.
Identification of isolates
Gram staining, morphological characteristics, and biochemical tests were used for the identification of bacterial pathogens while morphological characteristics, hyphae, and lactophenol cotton blue mount were used to identify the fungal pathogens.[8]
For the recovery and identification of parasites, the physiological saline solution obtained after shaking the flies in a sterile container was transferred into a test tube and centrifuged. The deposit was examined first by adding few drops of physiological saline and second by adding iodine solution.
Antibiotic susceptibility test
The antibiotic susceptibility of the isolates was tested against the following antibiotics: Ofloxacin (OFL) 10 µg, gentamicin (CN) 10 µg, amoxicillin/clavulanate (AMC) 30 µg, pefloxacin (PEF) 10 µg, cotrimoxazole (COT) 30 µg, streptomycin 30 µg, cephalexin 30 µg, ceftriaxone (CRO) 10 µg, and ampicillin (AMP) 30 µg. Antibiotic sensitivity pattern was determined by disc diffusion method.[9] A colony of the test organism was picked with a sterile wire loop and immersed in peptone water. The turbidity of the suspension was compared against a reference 0.5 McFarland tube. The suspension of the organism was streaked on the entire plate of nutrient agar and the antibiotic disc was placed on the plate using forceps. The plates were incubated at 37°C for 24 h.
Sensitivity pattern was determined by measuring the diameter of the zones of inhibition with a calibrated ruler and interpreted according to standard guidelines for Clinical and Laboratory Standards Institute criteria.[10]
Data analysis
Simple percentages were used for comparisons in the study, except for the evaluation of bacterial load where standard deviations and means were employed.
RESULTS
Of the 150 flies collected from different sites in Umuahia metropolis, the number collected from the different sites was as follows: Market 40 (26.7%), refuse dump 30 (20.0%), broken sewage 30 (20.0%), restaurants 30 (20.0%), and clinic 20 (13.3%). The results are shown in Figure 1.
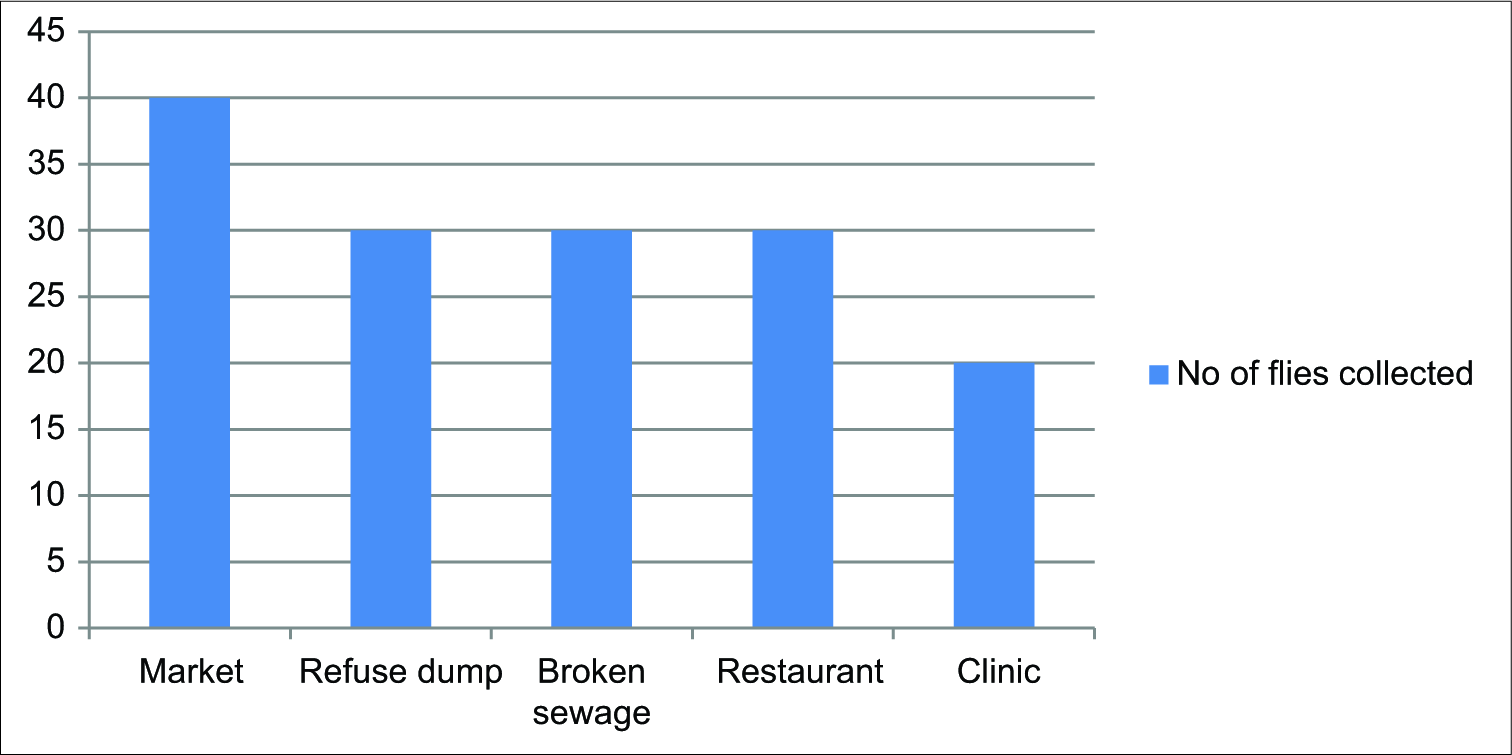
- Sites of collection and number of houseflies from each site.
The following bacterial isolates were identified; Table 1 Escherichia coli 35 (22.3%), Klebsiella spp. 25 (16.6%), Staphylococcus aureus 23 (14.7%), Proteus vulgaris 16 (10.2%), Proteus mirabilis 14 (8.92%), Salmonella spp. 11 (7.0%), Shigella spp. 10 (6.4%), Pseudomonas aeruginosa 9 (5.73%), Staphylococcus epidermidis 7 (4.5%), Enterococcus faecalis 5 (3.2%), and Streptococcus spp. 1 (0.64%).
| Bacterial isolates | Number (%) of isolates from | |||||
|---|---|---|---|---|---|---|
| Market | Refuse | Broken sewage | Restaurant | Clinic | Total | |
| Escherichia coli | 6 (26.09) | 8 (22.22) | 9 (18.37) | 6 (22.22) | 2 (27.27) | 35 (22.29) |
| Proteus vulgaris | 4 (17.39) | 4 (11.11) | 0 (0) | 3 (11.11) | 5 (22.73) | 16 (10.19) |
| Proteus mirabilis | 3 (13.04) | 2 (5.56) | 7 (14.29) | 2 (7.41) | 0 (0) | 14 (8.92) |
| Klebsiella spp. | 3 (13.04) | 4 (11.11) | 8 (16.33) | 6 (22.22) | 5 (22.73) | 26 (16.56) |
| Pseudomonas aeruginosa | 0 (0) | 5 (13.89) | 4 (8.16) | 0 (0) | 0 (0) | 9 (5.73) |
| Salmonella spp. | 0 (0) | 3 (8.33) | 5 (10.20) | 3 (11.11) | 0 (0) | 11 (7.01) |
| Shigella spp. | 0 (0) | 3 (8.33) | 4 (8.16) | 3 (11.11) | 0 (0) | 10 (6.37) |
| Enterococcus faecalis | 0 (0) | 2 (5.56) | 3 (6.12) | 0 (0) | 0 (0) | 5 (3.18) |
| Staphylococcus aureus | 4 (17.39) | 5 (13.89) | 6 (12.24) | 4 (14.81) | 4 (18.18) | 23 (14.65) |
| Staphylococcus epidermidis | 2 (8.70) | 0 (0) | 3 (6.12) | 0 (0) | 2 (9.09) | 7 (4.46) |
| Streptococcus spp. | 1 (4.35) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 1 (0.64) |
| Total | 23 | 36 | 49 | 27 | 22 | 157 |
A total of 157 bacterial isolates were identified and the number of isolates identified in each site was as follows:
Market 23 (14.6%), refuse dump 36 (22.9%), broken sewage 49 (31.2%), restaurant 27 (17.2%), and clinic 22 (14.0%).
Table 2 shows the mean bacterial load and range of total viable count (cfu/ml) of bacterial isolates collected from all sites. The total viable count of the bacterial isolates based on the site of collection include market ([4.75 ± 2.80–10.1 ± 30.6] × 108 cfu/ml), refuse dump ([4.50 ± 0.37–11.0 ± 4.8] × 108 cfu/ml), broken sewage ([7.15 ± 1.00–12.9 ± 8.9] × 108 cfu/ml), restaurant ([3.75 ± 1.05–9.45 ± 2.5] × 108 cfu/ml), and clinic ([3.5 ± 0.9–8.35 ± 2.01] × 108 cfu/ml). This indicates that the counts from the broken sewage and refuse dump have greater values compared to that from other sites.
| Isolates | Market (cfu/ml) | Refuse (cfu/ml) | Broken sewage (cfu/ml) | Restaurant (cfu/ml) | Clinic (cfu/ml) |
|---|---|---|---|---|---|
| Escherichia coli | 6.00±1.31×105 | 15.00±3.58×105 | 12.00±3.46×105 | 5.00±3.74×105 | 6.00±2.16×105 |
| Proteus vulgaris | 5.00±1.31×105 | 4.00±2.00×105 | - | 3.00±1.41×105 | 5.00±0.82×105 |
| Proteus mirabilis | 3.00±1.69×105 | 3.00±1.79×105 | 6.00±2.34×105 | 1.00±2.26×105 | - |
| Klebsiella spp. | 2.00±1.41×105 | 4.00±2.83×105 | 12.00±2.83×105 | 4.00±2.10×105 | 6.00±2.16×105 |
| Staphylococcus aureus | 4.00±2.62×105 | 5.00±1.79×105 | 11.00±2.83×105 | 4.00±1.41×105 | 4.00±1.83×105 |
| Staphylococcus epidermidis | 2.00±1.69×105 | - | 3.00±3.16×105 | - | 2.00±1.83×105 |
| Streptococcus spp. | 1.00±1.07×104 | - | - | - | - |
| Pseudomonas spp. | - | 4.00±3.03×105 | 3.00±1.89×105 | - | - |
| Salmonella spp. | - | 3.00±2.37×104 | 6.00±4.52×105 | 3.00±2.16×105 | - |
| Shigella spp. | - | 2.00±1.26×105 | 4.00±2.10×105 | 3.00±1.45×105 | - |
| Enterococcus spp. | - | 2.00±1.26×104 | 2.00±1.41×105 | - | - |
| Range of total viable count | (4.75±2.80–10.1±30.6)×108 | (4.50±0.37–11.0±4.8)×108 | (7.15±1.00–12.9±8.9)×108 | (3.75±1.05–9.45±2.5)×108 | (3.5±0.9–8.35±2.01)×108 |
The frequency of the occurrence of the fungal isolates from the external surface of these flies with respect to their sites of collection is shown in Table 3. Sixty fungal pathogens were isolated, namely, Aspergillus spp. 17 (28.3%), Mucor spp. 13 (21.7%), Penicillium spp. 7 (11.7%), Rhizopus spp. 9 (15.0%), and Candida spp. 14 (15.6%).
| Fungi isolates | Number (%) of fungal isolates from | |||||
|---|---|---|---|---|---|---|
| Market | Refuse dump | Broken sewage | Restaurant | Clinic | Total | |
| Aspergillus spp. | 3 (27.27) | 4 (25.00) | 5 (29.41) | 3 (37.50) | 2 (25.00) | 17 (28.33) |
| Mucor spp. | 2 (18.18) | 4 (25.00) | 3 (17.65) | 2 (25.00) | 2 (25.00) | 13 (21.67) |
| Penicillium spp. | 1 (9.09) | 2 (12.50) | 3 (17.65) | 0 (0) | 1 (12.50) | 7 (11.67) |
| Rhizopus spp. | 2 (18.18) | 3 (18.75) | 2 (11.76) | 1 (12.50) | 1 (12.50) | 9 (15.00) |
| Candida spp. | 3 (27.27) | 3 (18.75) | 4 (23.52) | 2 (25.00) | 2 (25.00) | 14 (15.56) |
| Total | 11 | 16 | 17 | 8 | 8 | 60 |
Table 4 shows the frequency of the occurrence of parasites from different sites. Fifty-five parasites were found and they were as follows: Endolimax nana 17 (30.9%), Giardia lamblia 13 (23.6%), Ascaris lumbricoides 7 (12.7%), and Entamoeba histolytica 18 (32.73).
| Parasites | Number (%) of isolates from | |||||
|---|---|---|---|---|---|---|
| Market | Refuse dump | Broken sewage Restaurant | Clinic | Total | ||
| Endolimax nana cyst | 3 (42.85) | 5 (35.71) | 8 (36.36) | 0 (0) | 1 (12.50) | 17 (30.91) |
| Giardia lamblia cyst | 1 (14.28) | 3 (21.42) | 5 (22.73) | 2 (50.00) | 2 (25.00) | 13 (23.64) |
| Entamoeba histolytica cyst | 2 (28.57) | 4 (28.57) | 6 (27.27) | 1 (25.00) | 5 (62.50) | 18 (32.73) |
| Ascaris lumbricoides ova | 1 (14.28) | 2 (14.28) | 3 (13.64) | 1 (25.00) | 0 (0) | 7 (12.72) |
| 7 | 14 | 22 | 4 | 8 | 55 | |
The antibiotic sensitivity patterns of the non-healthcare- related isolates (i.e., isolates from market, refuse dump, broken sewage, and restaurant) are shown in Table 5 while the antibiotic sensitivity pattern of the isolates obtained from cockroaches from health-care environment (i.e., isolates from the clinic) is shown in Table 6. These isolates showed high level of resistance to AMP and COT while OFL and CRO showed encouraging results.
| Bacteria | Number of tested | Number (%) of isolates sensitive to | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| OFX | PEF | CRO | AMC | CN | S | CEP | COT | AMP | ||
| Escherichia coli | 29 | 25 (86.2) | 17 (58.6) | 26 (89.7) | 23 (79.3) | 18 (62.1) | 4 (13.8) | 9 (31.0) | 3 (10.3) | 8 (27.5) |
| Proteus vulgaris | 11 | 10 (90.9) | 7 (63.6) | 9 (81.8) | 7 (63.6) | 11 (100) | 10 (62.5) | 2 (18.1) | 3 (27.3) | 7 (63.6) |
| Proteus mirabilis | 14 | 10 (71.4) | 7 (43.8) | 12 (85.7) | 7 (50.0) | 10 (71.4) | 11 (78.6) | 3 (21.4) | 3 (21.4) | 6 (42.9) |
| Klebsiella spp. | 21 | 13 (61.9) | 6 (28.6) | 16 (76.2) | 12 (57.1) | 12 (57.1) | 10 (47.6) | 12 (57.1) | 5 (23.9) | 5 (23.9) |
| Pseudomonas aeruginosa | 9 | 5 (53.6) | 4 (44.4) | 7 (77.8) | 0 (0) | 6 (66.7) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Salmonella spp. | 11 | 7 (63.3) | 7 (63.6) | 8 (72.7) | 0 (0) | 7 (63.6) | 2 (18.2) | 7 (63.6) | 1 (9.1) | 0 (0) |
| Shigella spp. | 9 | 7 (70.0) | 5 (50.0) | 6 (66.7) | 4 (44.4) | 9 (90.0) | 3 (33.3) | 3 (33.3) | 2 (22.2) | 1 (11.1) |
| Staphylococcus aureus | 19 | 14 (73.7) | 18 (94.7) | 16 (84.2) | 12 (63.2) | 10 (52.6) | 0 (0) | 10 (52.6) | 8 (42.1) | 6 (31.6) |
| Staphylococcus epidermidis | 5 | 4 (80.0) | 0 (0) | 2 (40.0) | 0 (0) | 2 (40.0) | 0 (0) | 2 (40.0) | 3 (60.0) | 0 (0) |
| Streptococcus spp. | 1 | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Enterococcus faecalis | 5 | 3 (60.0) | 0 (0) | 0 (0) | 0 (0) | 2 (40.0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
OFX: Ofloxacin, PEF: Pefloxacin, CRO: Ceftriaxone, AMC: Amoxicillin/clavulanate, CN: Gentamicin, S: Streptomycin, CEP: Cephalexin, COT: Cotrimoxazole, AMP: Ampicillin
| Isolates | Number of tested | Number (%) of isolates sensitive to | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| OFX | PEF | CRO | AMC | CN | S | CEP | COT | AMP | ||
| Escherichia coli | 6 | 4 (66.0) | 3 (50.0) | 2 (33.3) | 1 (16.7) | 2 (33.3) | 1 (16.7) | 1 (16.7) | 0 (0) | 0 (0) |
| Proteus vulgaris | 5 | 2 (40.0) | 1 (20.0) | 3 (60.0) | 2 (40.0) | 2 (40.0) | 2 (40.0) | 0 (0) | 0 (0) | 0 (0) |
| Klebsiella spp. | 5 | 1 (20.0) | 2 (40.0) | 1 (20.0) | 0 (0) | 3 (60.0) | 1 (25.0) | 0 (0) | 0 (0) | 0 (0) |
| Staphylococcus aureus | 4 | 1 (25.0) | 0 (0) | 2 (50.0) | 0 (0) | 2 (50.0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Staphylococcus epidermidis | 2 | 1 (50.0) | 0 (0) | 1 (50.0) | 0 (0) | 1 (50.0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
OFX: Ofloxacin, PEF: Pefloxacin, CRO: Ceftriaxone, AMC: Amoxicillin/clavulanate, CN: Gentamicin, S: Streptomycin, CEP: Cephalexin, COT: Cotrimoxazole, AMP: Ampicillin
DISCUSSION
The entomology of houseflies has established them as notable vector of diseases. They are common around households, garbage, human, and animal excreta.[11]
In this study, 11 bacteria genera were isolated from the external surface of houseflies collected from sites previously mentioned, indicating that the external organs of M. domestica (legs, wings, and mouthparts) constitute a large source of bacteria which are in agreement with the report from Tangier, Morocco.[12] The isolates which are bacteria genera of medical importance include E. coli, S. aureus, Pseudomonas spp., Klebsiella spp., Salmonella spp., and Shigella spp. This observation is in accordance with the findings of other researchers.[6,13,14]
The distribution of isolates showed that E. coli had the highest frequency of occurrence which is in line with the findings of other researchers.[14,15] The presence of E. coli can only signify fecal contamination which is easily carried by flies.[1]
The bacterial load observed in this study was highest from flies collected from broken sewage followed by isolates from flies recovered from refuse dumps. It is, however, not surprising due to the high level of fecal matter and organic debris associated with these sites.
The presence of Salmonella spp. and Shigella spp. isolated from this study and also found in another study from Uturu, Nigeria,[16] portends great danger because they could cause severe gastroenteritis which could eventually lead to death if not properly managed.
The isolation of Aspergillus spp. and Penicillin spp. from this study agrees with the findings from other researchers.[2] Infections with Aspergillus flavus and related molds which frequently contaminate corns, grains, and other foods portends danger and are of public health significance due to the production of aflatoxins and could be transmitted by houseflies. Another report also in Umuahia, Nigeria,[17] observed that cysts of E. histolytica and G. lamblia and ova of A. lumbricoides have high frequency of occurrence on houseflies that are found around broken sewage. This compares favorably with the reports from this study.
The antibiotic sensitivity pattern of the bacterial isolates from flies collected from the health-care environments exhibited resistance to most of the antibiotics used. An earlier report[6] from a similar study in a health-care setting established the multiresistance profiles of the bacterial isolates from the environment. However, the rate of antibiotic susceptibility of the bacterial isolates from flies recovered from other sites in this study is at variance with the findings from earlier report.
CONCLUSION
The presence of houseflies indicates sanitary deficiency and unhygienic conditions. The findings established from the results of this study have established that houseflies can be efficient vectors for the mechanical transmission of multidrug-resistant diseases causing organisms, especially from a health-care environment. The diseases transmitted by these houseflies could pose serious health risks to children, elderly people, and immunocompromised individuals. The presence of houseflies indicates sanitary deficiency and unhygienic conditions that deserve prompt attention to prevent the spread of superbugs within the community.
Recommendation
The study recommends good sanitation practices, adequate waste disposal system, and the elimination possible breeding sites for houseflies in homes, clinics, and offices. Since the major transfer substrate for bacteria, fungi, or parasites is food, proper heating and covering of food before consumption are also good practice.
Limitation
Antisera were not available for use to confirm the pathogenic serotypes for E. coli gastroenteritis.
Acknowledgment
We thank members of staff in both zoology and microbiology departments for their kind assistance during the period of this research.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Pathogenic microorganisms associated with houseflies within Uyo metropolis during the wet season. Researcher. 2012;4:37-41.
- [Google Scholar]
- Bacterial fauna from the house fly, Musca domestica (L) Trop Biomed. 2005;22:225-31.
- [Google Scholar]
- Isolation of Salmonella enterica serovar enteritidis from houseflies (Musca domestica) found in rooms containing Salmonella serovar enteritidis challenged hens. Appl Environ Microbiol. 2007;73:6030-5.
- [CrossRef] [PubMed] [Google Scholar]
- Review article: Is Helicobacter pylori transmitted by the gastro-oral route? Aliment Pharmacol Ther. 1995;9:585-8.
- [CrossRef] [PubMed] [Google Scholar]
- Transmission of Bacterial pathogens by housefly Musca domestica vicina. Am J Res Commun. 2013;1:1-12.
- [Google Scholar]
- Isolation and identification of Bacterial isolates from houseflies in Sulaymanya city. Eng Technol J. 2013;31:24-33.
- [Google Scholar]
- The role of non-biting flies in the epidemiology of human infectious diseases. Microbes Infect. 2011;3:231-5.
- [CrossRef] [Google Scholar]
- Medical Laboratory manual in Tropical Countries. In: Microbiology. Vol 2. London: Tropical Health Technology; 2006. p. :124-6.
- [CrossRef] [Google Scholar]
- Antibiotic susceptibility testing by a standard single disc method. Am J Clin Pathol. 1966;45:493-6.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical and Laboratory Standards Institute. In: Performance Standards for Antimicrobial Susceptibility Testing; Twenty-second Informational Supplement (M100-S22). Vol 32. Wayne, PA: Clinical and Laboratory Standards Institute; 2012. p. :6030-5.
- [Google Scholar]
- The role of cockroaches and flies in mechanical transmission of medical important parasites. J Entomol. 2011;3:98-104.
- [Google Scholar]
- Antibiotic resistance patterns of Bacterial isolates strains isolated from Periplaneta american and Musca domestica in Tangier, Morocco. J Infect Dev Ctries. 2010;4:194-201.
- [CrossRef] [PubMed] [Google Scholar]
- Identification of bacteria which are possibly transmitted by Musca domestica (dipthera Muscidae) in the region of Ahvaz, SW Iran. Jundishapur J Microbiol. 2008;1:28-31.
- [Google Scholar]
- Isolation of pathogenic Bacteria on the housefly Musca domestica L. (diptheria Muscidae) body surface in Ahwaz hospital Southwestern Iran. Asian Pac J Trop Biomed 2012:S1116-9.
- [CrossRef] [Google Scholar]
- Wild Florida flies (Musca domestica) as carriers of pathogenic Bacteria. Fla Entomol. 2010;93:218-23.
- [CrossRef] [Google Scholar]
- Isolation of Salmonella and Shigella species from house flies (Musca domestica) in Uturu, Nigeria. Afr J Biotechnol. 2006;5:1090-1.
- [Google Scholar]
- Parasitic load on Musca domestica (dipthera Muscidae) from different synanthropic environments in Umuahia metropolis. J Public Health Epidemiol. 2013;5:309-12.
- [Google Scholar]






