Translate this page into:
Atypical presentations of orbital cellulitis in an 11-year-old boy

-
Received: ,
Accepted: ,
How to cite this article: Oraegbunam NH, Ezeh EI, Etiowo N, Ezeh RN. Atypical presentations of orbital cellulitis in an 11-year-old boy. Calabar J Health Sci 2020;4(1):40-3.
Abstract
Spontaneous acute-onset proptosis accompanied with inflammatory signs in children is commonly caused by orbital cellulitis/abscess; however, the clinician should always be alert to the possibility of other causes such as neoplastic: Orbital rhabdomyosarcoma (RMS), traumatic, and iatrogenic factors. This is a case report of an 11-year-old boy presenting with an acute-onset non-axial proptosis of the left eye without a history of trauma, sinus disease, or systemic infection. Our clinical differential diagnosis included orbital cellulitis and orbital RMS. However, the final diagnosis was orbital cellulitis with abscess. The purpose of the study was to report a case of “cold” orbital abscess that clinically mimics orbital RMS. An 11-year-old boy presented with a 2 weeks history of painless, rapid-onset non-axial proptosis in the left eye. It was associated with periorbital edema, and mild conjunctival hyperemia. There was no preceding or associated history of fever, trauma, upper respiratory tract infection, sinusitis, or immunosuppression. An initial clinical diagnosis of RMS, with orbital cellulitis as a differential diagnosis, was made delaying commencement of antibiotic therapy. Following the drainage of greenish tinged purulent matter growing Staphylococcus aureus on culture, the diagnosis of orbital cellulitis with abscess was made. A broad-spectrum antibiotics and subsequent adjunct anti-inflammatory therapy yielded excellent clinical resolution. The case demonstrates the pitfalls/challenges in differentiating orbital space occupying lesions manifesting with inflammatory features on the basis of clinical findings alone. This is particularly relevant in a busy triage ophthalmic clinic in a low resource environment without easy access to any form of orbital imaging. In addition, the case highlights that green tinged purulent matter can be found in infections from a myriad of organisms aside from the popularly known Pseudomonas aeruginosa.
Keywords
Proptosis
Orbital
Abscess
Cellulitis
Rhabdomyosarcoma
INTRODUCTION
Any purulent collection in the orbit is known as an orbital abscess. It typically follows an infection of the orbital soft tissues (orbital cellulitis). Classic clinical findings include fever, erythema, proptosis, chemosis, ptosis, restriction of and pain with ocular movement, reduced visual acuity, and an afferent pupillary defect.[1-4] Orbital cellulitis/abscess is the most common cause of acute-onset proptosis with inflammatory signs in children.[3,4] The differential diagnosis of orbital cellulitis includes trauma which may occur without a reliable history, non-specific orbital inflammation, and benign orbital tumors such as lymphatic malformation and hemangioma, and malignant tumors such as rhabdomyosarcoma (RMS), leukemia, and metastases. RMS is a particularly important differential because of its classic picture of sudden onset and rapid progression of unilateral proptosis and globe displacement. It is associated with marked ocular adnexal inflammatory response in 60% of cases therefore mimicking orbital cellulitis.[5-7] RMS is the most common primary orbital malignancy in children.[1,5,7]
We report a case of orbital cellulitis with abscess that mimics RMS in an 11-year old boy with rapid-onset unilateral proptosis.
CASE PRESENTATION
An 11-year old, primary six, male pupil presented, accompanied by his widowed mum, to Triage section of the Eye clinic of University of Calabar Teaching Hospital with a 2 weeks history of spontaneous, sudden onset of a rapidly progressing painless mass at the inferior part of the left orbit, associated redness, and slight forward and upward displacement of the eyeball. There was no history of cough, catarrh, sore throat, facial heaviness, oral pains, fever, weight loss, or trauma to the eye. He admitted to feeling a mild pressure sensation in the left eye, but no reduction in his vision. He took only paracetamol before presentation and categorically denied use of traditional eye medication. He has no significant medical history. His immunization history was up to date.
Examination revealed a calm child in no obvious painful distress, afebrile (36.9°C), not pale, anicteric and had no preauricular or submandibular lymphadenopathy. The right eye was essentially normal. The left eye showed mildly edematous, non-tender eyelids with no differential warmth. The bulbar conjunctiva was moderately injected with yellowish, and necrotic area in the inferior fornix. A firm non-tender mass was felt occupying the inferior part of the anterior orbit. There was a mild proptosis and superior dystopia of 2 mm was noted. A mild restriction of ocular motility was noted on downgaze but double vision was not elicited. He had normal visual acuities, pupillary responses, and fundus examination. An initial clinical diagnosis of orbital RMS was made with orbital cellulitis as a differential diagnosis. He was admitted for joint management by the pediatric ophthalmology and oculoplastic teams. Baseline bloods (Full Blood Count with differentials, Liver Function Test, and Serum Electrolytes/Urea/Creatinine), and computerized tomography (CT) head, and orbit requested.
On review the next day by the teams, the findings were in keeping with the initial clinical findings. However, on palpation to determine the extent of the orbital mass, it inadvertently ruptured releasing copious amounts of green, non-foul smelling pus estimated at 4 ml [Figure 1].
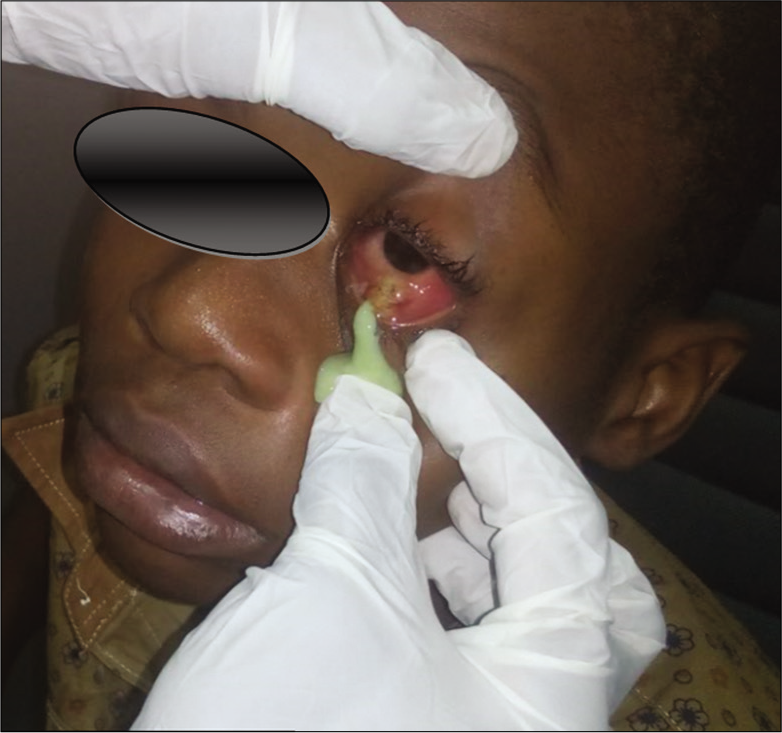
- Clinical photograph showing draining of greenish pus from left anterior orbit.
The pus was expressed till clear blood released, a sample of the purulent matter was sent for Microscopy/Culture/Sensitivity. A definitive diagnosis of orbital abscess was then made and he was commenced empirically on intravenous ceftriaxone 1 g daily, intravenous metronidazole 250 mg tds, and ointment chloramphenicol tds left eye for 1 week. Culture after 48 h yielded growth of Staphylococcus aureus only. Full blood count showed marginal elevation of white blood cell count. Requested orbito-cranial CT scan of the orbit was not done due to lack of finances.
He was discharged after 8 days on admission on tablets amoxicillin-clavulanic acid 375 mg bd and tablet prednisolone 20 mg daily for 2 weeks. At discharge, there was remarkable regression of the ocular redness, proptosis, globe displacement, and ocular motility.
Follow up 2 weeks after discharge, revealed a stable child with complete resolution, as the globe has recovered, proptosis and displacement disappeared and ocular motility became normal.
DISCUSSION
The clinical presentation of some orbital disease in children may occasionally be deceptive, particularly if atypical. Diagnosis of space-occupying lesions in the orbit is particularly challenging because the clinical manifestations are both non-specific and relatively limited. The problem of differential diagnosis in childhood is compounded by the fact that many different orbital processes may cause rapid onset of symptoms.[7] The differential diagnoses of rapidly progressing nontender or slightly tender orbital masses could be inflammatory, infectious, or neoplastic.[1,7]
Although children with orbital cellulitis may have varied presentations, the common symptoms include: Painful swelling of the upper and lower eyelid, bulging of the eyeball, reduction in the child’s ability to move the eyeball, pain on eye movement, chemosis, and fever.[2-4] Similarly, the classic clinical picture of RMS is that of a child with sudden onset and rapid progression of unilateral proptosis. There is often a marked adnexal response with edema and discoloration of the eyelids, interestingly, ocular, and orbital pains are less common presenting symptoms ranging from 10% to 20%. A mass may be palpable, particularly in the superonasal quadrant of the eyelid.[5-7] Hence, in children presenting with acute rapid-onset painless unilateral proptosis with signs of inflammation, the possibility of RMS should always be consider.[8,9] This was the consideration in our index patient who presented with sudden onset of a rapidly progressing painless mass at the inferior part of the left orbit, associated redness, and slight forward and upward displacement of the eyeball. There was a marked absence systemic signs such as fever and lethargy. It is however possible that the ingestion of paracetamol by our index patient may have masked the pain and fever. It is therefore pertinent to note that interventions by care givers or patients before presentation at health facility can remarkably alter the natural history and clinical features of a disease, thus misleading the clinician.
Acute, rapidly progressive proptosis with an absence of pain, fever, and features of sinus infection, upper respiratory tract infection or dental infection, should as presented in this index case raise a high suspicion of orbital RMS. However, based on the marginal leukocytosis, the purulent collection drained yielding bacterial growth and remarkable response to antibiotic therapy, it was no doubt a case of orbital cellulitis presenting as a relatively “cold” orbital abscess.
RMS is a highly malignant tumor, in which the tissue of origin is pluripotent mesenchyme.[10] It is the most common soft-tissue sarcoma of the head and neck in childhood and comprises 4% of all pediatric malignancies, with 10% of all cases occurring in the orbit.[10,11] The average age of onset is 8–10 years.[1] Orbital RMS is most commonly superonasal in location especially with embryonal RMS but an inferior location is common for alveolar type.[12] Clinical history may provide for suspicion of the differential diagnoses for RMS such as orbital cellulitis, lymphangioma, idiopathic orbital inflammation, dermoid cyst, hemangioma, Langerhans cell histiocytosis (eosinophilic granuloma), sarcomas, metastatic neuroblastoma, and lymphoma.[11] Imaging plays an important role in the differentiation of these conditions. Biopsy should be performed if RMS is suspected based on clinical and radiological findings.[11,12]
This case illustrates the difficulty in differentiating orbital infection from orbital RMS on the basis of clinical findings. In our patient, it was largely due to the absence of typical periocular and systemic features seen with orbital cellulitis thereby mimicking orbital RMS. It is therefore pertinent that the clinician should always be aware of the low reliability of the common clinical features of orbital disease (proptosis/ dystopia; swelling or discoloration of the eyelid; and palpable subcutaneous mass) in making diagnosis. A combination of clinical, laboratory, orbital imaging, and biopsy may sometimes require for definitive diagnosis in a child with acute-onset proptosis with inflammatory signs.[8,9] Hegde and Sundar[2] posited that although the diagnosis of orbital cellulitis is a clinical one, sino-orbital imaging, usually by CT scan is often required to identify underlying sinusitis or when suspected confirm the presence and location of a subperiosteal abscess (SPA), rule out unsuspected foreign bodies, or rarely even masquerade syndromes.
The direction of the proptosis/dystopia depends on the location of the primary orbital lesion which tends to maintain in one space except for malignant lesions. Common locations for the abscess formation are adjacent to opacified paranasal sinuses. Specifically, frequent locations for the development of SPAs include the medial orbital wall and the orbital floor, given the thin medial wall adjacent to the ethmoid sinus and the thin orbital floor above the maxillary sinus, respectively.[13] Our index patient had a mild proptosis with superior dystopia in keeping with a mass in the anteroinferor orbit possibly restricted to the anterior subtenon or peripheral orbital space [Figure 2]. This location explains the ease of drainage of the abscess to the ocular surface. However, clinicians should be mindful of the fact that alveolar RMS is commonly located in the inferior orbit.
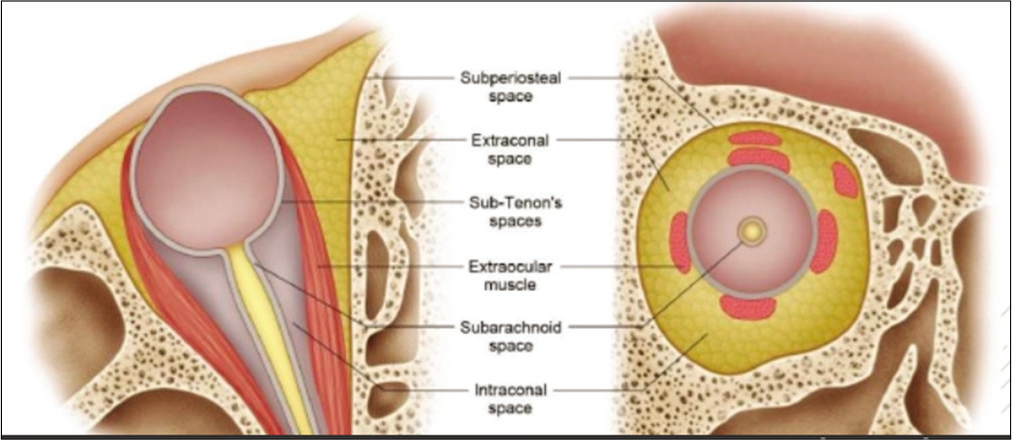
- Surgical spaces in the orbit.[1]
Another atypical feature in the index patient is the isolation of S. aureus from green tinged pus. The typical pathogens associated with acute sinusitis, including Streptococcus pneumoniae, Streptococcus pyogenes, S. aureus, Haemophilus influenzae, Moraxella catarrhalis, non-hemolytic streptococci (including the Streptococcus milleri group), and some anaerobic strains such as Bacteroides, Peptostreptococcus, and Fusobacterium are common causes of orbital cellulitis.[3,14-16] S. aureus isolated in our patient has been reported as the causative agent in about 30% of patients.[16] Our index patient had brightly green non foul smelling, purulent discharge; however, the usual coloration of purulent matter from S. aureus infection is whitish-yellow.[16] This is misleading as greenish pus is in general attributed to Pseudomonas aeruginosa. The greenish color of foul-smelling pus is found that P. aeruginosa infections are a result of the bacterial pigment pyocyanin which the organism produces. It has, however, been documented that green tinged pus is also produced in infections in which large amounts of myeloperoxidase (MPO), an intensely green antibacterial protein, is produced by neutrophils, lymphocytes, monocytes, and macrophages.[17,18] MPO exerts potent and broad-spectrum microbicidal action against Gram-positive (S. aureus) and Gram-negative bacteria (Escherichia coli and P. aeruginosa), as well as yeast and fungi. Release of MPO at the sites of inflammation leads to the production of reactive oxygen species and reactive nitrogen species which are toxic to several microorganisms and play an important role in the immune system.[18]
CONCLUSION
Orbital cellulitis/abscess may clinically mimic orbital RMS. Clinicians should entertain the possibility of atypical forms of clinical presentation and the need for a special attention on children presenting with an acute rapid-onset progressive proptosis. Broad mind-set with high index of suspicion in the management of pediatric ophthalmic patients in a busy ophthalmic clinic is paramount, particularly in low resource settings.
Notably, green tinged pus could be produced in microbial infections, in which large amounts of MPO are elaborated by white blood cells.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Section 07: Orbit, eyelids and lacrimal system In: Cantor LB, Rapuano CJ, Cioffi GA, eds. 2016-2017 Basic and Clinical Science Course. San fransisco: American Academy of Opthalmology; 2016. p. :70-2.
- [Google Scholar]
- Orbital cellulitis in children. Pediatr Infect Dis J. 2006;25:695-9.
- [CrossRef] [PubMed] [Google Scholar]
- Orbital cellulitis in children. Int J Ophthalmol Clin. 2001;41:71-86.
- [CrossRef] [PubMed] [Google Scholar]
- Orbital space occupying lesion in Indian children. J Pediatr Ophthalmol Strabismu. 2007;44:106-11.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical, ultrasonographic and CT evaluation of orbital rhabdomyosarcoma with management. Indian J Ophthalmol. 1991;39:129-31.
- [Google Scholar]
- Section 06: Pediatric ophthalmology and strabismus In: Cantor LB, Rapuano CJ, Cioffi GA, eds. 2016-2017 Basic and Clinical Science Course. San Fransisco: American Academy of Ophthalmology; 2016. p. :313-4.
- [Google Scholar]
- Orbital cellulitis clinically mimicking rhabdomyosarcoma. Int Med Case Rep J. 2019;12:285-9.
- [CrossRef] [PubMed] [Google Scholar]
- Neoplasia In: Diseases of the Orbit: A Multidisciplinary Approach. Philadelphia, PA: Lippincott Williams and Wilkins; 2003. p. :262-8.
- [Google Scholar]
- Rhabdomyosarcoma: Review for the ophthalmologist. Surv Ophthalmol. 2003;48:39-57.
- [CrossRef] [Google Scholar]
- Orbital rhabdomyosarcomas: A review. Saudi J Ophthalmol. 2013;27:167-75.
- [CrossRef] [PubMed] [Google Scholar]
- Management of preseptal and orbital cellulitis. Saudi J Ophthalmol. 2011;25:21-9.
- [CrossRef] [PubMed] [Google Scholar]
- Paediatric pre-and post-septal peri-orbital infections are different diseases. A retrospective review of 262 cases. Int J Pediatr Otorhinolaryngol. 2008;72:377-83.
- [CrossRef] [PubMed] [Google Scholar]
- Microbiology and antimicrobial treatment of orbital and intracranial complications of sinusitis in children and their management. Int J Pediatr Otorhinolaryngol. 2009;73:1183-6.
- [CrossRef] [PubMed] [Google Scholar]
- Sinogenic orbital and subperiosteal abscesses: Microbiology and methicillin-resistant Staphylococcus aureus incidence. Otolaryngol Head Neck Surg. 2010;143:392-6.
- [CrossRef] [PubMed] [Google Scholar]
- Wikipedia, The Free Encyclopedia. 2019. Available from: https://www.en.wikipedia.org/w/index.php?title=Pus&oldid=924659971 [Last accessed on 2019 Nov 15]
- [Google Scholar]
- Myeloperoxidase as an active disease biomarker: Recent biochemical and pathological perspectives. Med Sci (Basel). 2018;6:33.
- [CrossRef] [Google Scholar]






